
Researchers have discovered an antibody able to neutralize all known variants of SARS-CoV-2, the virus that causes COVID-19, as well as distantly related SARS-like coronaviruses that infect other animals.
As part of a new study on hybrid immunity to the virus, the large, multi-institution research team led by The University of Texas at Austin discovered and isolated a broadly neutralizing plasma antibody, called SC27, from a single patient. Using technology developed over several years of research into antibody response, the team led by UT engineers and scientists obtained the exact molecular sequence of the antibody, opening the possibility of manufacturing it on a larger scale for future treatments.
“The discovery of SC27, and other antibodies like it in the future, will help us better protect the population against current and future COVID variants,” said Jason Lavinder, a research assistant professor in the Cockrell School of Engineering’s McKetta Department of Chemical Engineering and one of the leaders of the new research, which was recently published in Cell Reports Medicine.
During the more than four years since the discovery of COVID-19, the virus that causes it has rapidly evolved. Each new variant has displayed different characteristics, many of which made them more resistant to vaccines and other treatments.
Protective antibodies bind to a part of the virus called the spike protein that acts as an anchor point for the virus to attach to and infect the cells in the body. By blocking the spike protein, the antibodies prevent this interaction and, therefore, also prevent infection.
SC27 recognized the different characteristics of the spike proteins in the many COVID variants. Fellow UT researchers, who were the first to decode the structure of the original spike protein and paved the way for vaccines and other treatments, verified SC27’s capabilities.
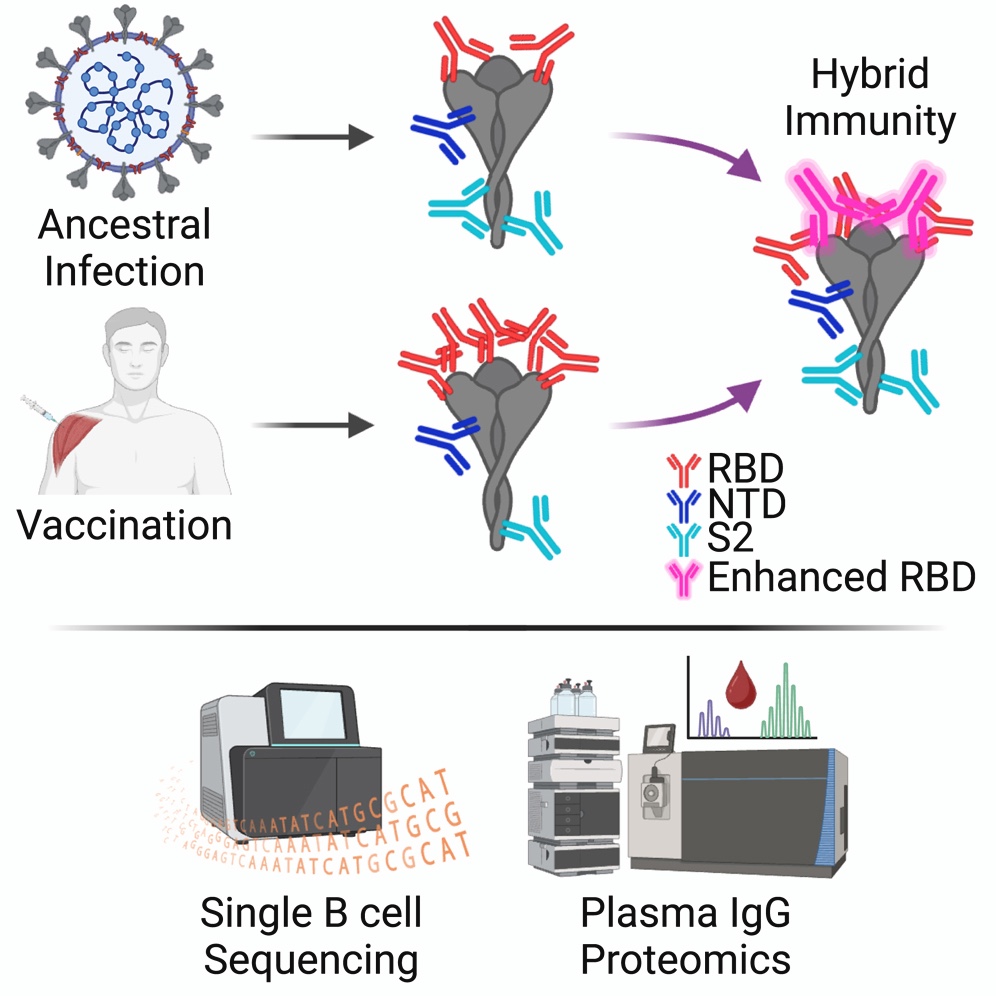
The technology used to isolate the antibody, termed Ig-Seq, gives researchers a closer look at the antibody response to infection and vaccination using a combination of single-cell DNA sequencing and proteomics.
“One goal of this research, and vaccinology in general, is to work toward a universal vaccine that can generate antibodies and create an immune response with broad protection to a rapidly mutating virus,” said Will Voss, a recent Ph.D. graduate in cell and molecular biology in UT’s College of Natural Sciences, who co-led the study.
In addition to the discovery of this antibody, the research found that hybrid immunity — a combination of both infection and vaccination — offers increased antibody-based protection against future exposure compared with infection or vaccination alone.
The work comes amid another summer COVID spike. This trend shows that while the worst of the pandemic may have passed, there’s still a need for innovative solutions to help people avoid and treat the virus.
The researchers have filed a patent application for SC27. Other members of the team from UT are Jason McLellan, Patrick O. Byrne, Sean A. Knudson, Douglas R. Townsend, Jessica Kain and Yimin Huang of the Department of Molecular Biosciences; George Georgiou, Ed Satterwhite and Allison Seeger of the McKetta Department of Chemical Engineering; Jeffrey M. Marchioni of the Department of Biomedical Engineering; and Chelsea Paresi of the Department of Chemistry. Team members from other institutions include Greg Ippolito of the Texas Biomedical Research Institute; Ralph S. Baric, Michael A. Mallory, John M. Powers, Sarah R. Leist, Jennifer E. Munt and Trevor Scobey of the University of North Carolina at Chapel Hill’s Department of Epidemiology; Izabella N. Castillo, Melissa Mattocks and Premkumar Lakshmanane of UNC’s Department of Microbiology and Immunology; and Bernadeta Dadonaite and Jesse D. Bloom of Fred Hutchinson Cancer Center. The research team received funding from the National Institutes of Health and the Bill & Melinda Gates Foundation.
For more information, contact: Nat Levy, Cockrell School of Engineering,






